Collaborate with Us
We welcome the potential to use our resources to advance biomedical science in areas ranging from basic laboratory to clinical therapeutics to clinical epidemiology, and other aspects of patient-oriented clinical investigation. We invite interested researchers to consider collaborating with us. There are multiple ways to do that – perhaps you’re interested in becoming a clinical site for one of our multicenter observational studies. Alternately, you may want to propose a substudy in our cohorts, or perform analyses using the clinical, imaging, and biomarker data created through our studies.
To propose a substudy for our cohorts, please contact Dr. Boxer with a short (one-paragraph) summary of your proposal. If you are interested in joining ALLFTD or 4RTNI as clinical sites, please contact Dr. Heuer to obtain a feasibility survey. We have limited financial resources for new sites, but are also excited to expand our networks.
Data from ALLFTD and 4RTNI are available to researchers and we encourage their use. Data and resource sharing occurs jointly for the studies and is managed through ALLFTD. The data request process is outlined at allftd.org/data. Biospecimens are available through NCRAD. MRIs for ALLFTD will be available for sharing in late 2022 following additional deidentification procedures. 4RTNI-2 also provides tau PET imaging and MRIs; these data may be available by request through the Laboratory of Neuroimaging (LONI). Please contact Dr. Heuer for details on receiving 4RTNI-2 images.
The flow of data requests through ALLFTD is shown here.
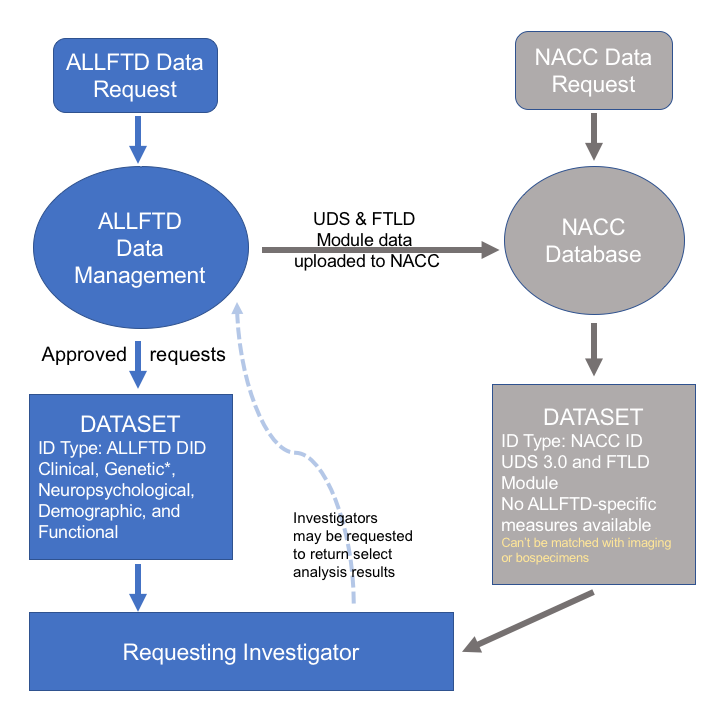
Please review the ALLFTD Data Sharing and Publication policy, and refer to the ALLFTD Data Guide and ALLFTD Data Display Guidelines for more information. We provide global scores for the CDR+NACC FTLD. These are calculated according to the rules shown in the FTLD-CDR Scoring Guide. We strongly recommend the use of the FTLD-CDR over the standard CDR for these data.
To request clinical data from ALLFTD/4RTNI, use the Data Request Form. Datasets are provided from the most recent quarterly data freeze, and requests are reviewed at least quarterly, often more frequently. Please email [email protected] with any questions. For biospecimen requests, please complete the Biospecimen Request Form. Biospecimen requests are reviewed on a bimonthly schedule.